Beskriver hvordan pustemønster er en grunnleggende aspekt av panikkangst og at pusteteknikker for å øke CO2 ved hjelpe av Capnografisk biofeedback virker terapeutisk med svært gode resultater. Etter 12 mnd rapportert 68% at de var blitt fri fra panikkangst og hele 96% var «mye bedre».
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2890048/
«Significant improvements (in PD severity, agoraphobic avoidance, anxiety sensitivity, disability, and respiratory measures) were seen in treated but not untreated patients, with moderate to large effect sizes. Improvements were maintained at follow-up. »
«That such training will result in higher levels of pCO2 cannot be taken for granted, since the usual instruction to breathe slowly can actually lead to decreases in pCO2 (Meuret et al., 2003; Ley, 1991), probably because of deeper individual breaths (higher tidal volumes) stimulated by feelings of suffocation.»
«Klein’s suffocation alarm hypothesis (Klein, 1993), for example, suggests that both panic attacks and the consistent respiratory abnormalities seen in panic patients may be due to hypersensitive, medullary carbon dioxide (CO2) detectors. »
The treatment had five major components: (a) educating patients about the role of breathing in the etiology and maintenance of PD, (b) directing their attention to potentially problematic respiratory patterns, particularly those observed during the extended physiological monitoring, (c) having them perform different breathing maneuvers with capnometer feedback to experience how changes in breathing affect physiology, symptoms, and mood, (d) teaching them ways to simultaneously control pCO2 level and RR (e) and having them practice breathing exercises daily.
Individual training exercises, to be performed twice- daily for 17-min, at home or elsewhere, consisted of three parts: (a) a baseline period (baseline), during which patients sat quietly with their eyes closed for 2-min, (b) a 10-min paced breathing period (paced) during which patients breathed in synchrony with tones while occasionally checking their pCO2 and RR on a feedback device, and (c) a 5-min breathing period without pacing tones during which patients were to maintain their previously paced RR and pCO2 level using the feedback device (transfer).
In addition, patients were provided with a pocket-sized tape player and audiotapes with instructions and pacing tones for their exercises. The tones were set to correspond to a RR of 13 breaths per minute in the first week, and rates of 11, 9, and 6 breaths per minute in successive following weeks.
During the maintenance period (between 2 and12-month FU) nine of the patients taking psychotropic medication had reduced their doses or discontinued medication altogether; four patients initiated alternative (n=3, relaxation, yoga, spiritual guidance) or psychological treatment (n=1, cognitive therapy).
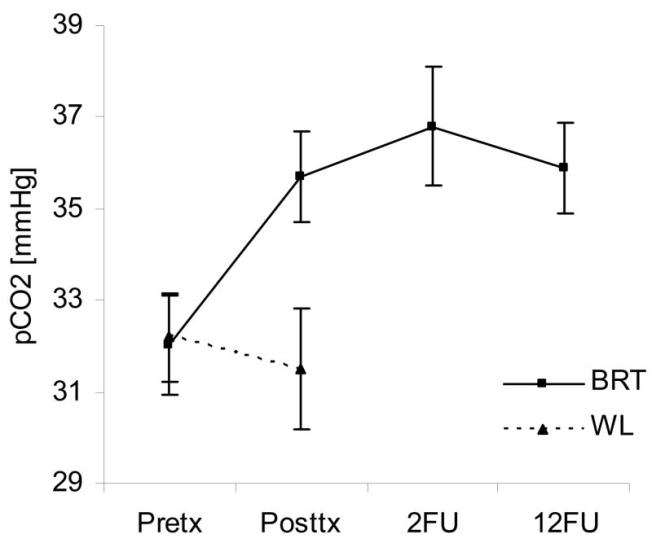
Post-hoc tests showed that pCO2 dropped significantly below baseline level during paced breathing and transfer in week 1 (34.7 and 34.5 mmHg). It reached the highest levels (around 38 mmHg) for all three phases during week 4.
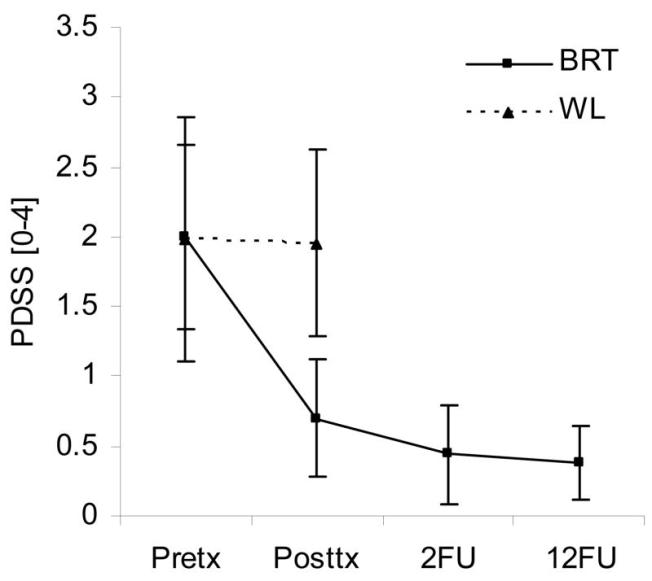
«At posttreatment 40% had experienced no further panic attack during the four week period. At 2-month follow-up 62% had experienced no further panic attack since the end of treatment and 68% were panic-free at 12-month follow-up. Eighty-eight percent at 2-month follow-up and 96% at 12-month follow-up were either “much improved” or “very much improved” »
The results of this study suggest that a new, brief, capnometry-assisted breathing therapy (BRT), which specifically teaches patients to raise pCO2 levels by regular slow and shallow breathing, can be therapeutic in PD. Significant improvements were seen in treated but not untreated patients, with respect to PD severity, agoraphobic avoidance, anxiety sensitivity, disability, and respiratory measures. Psychological measures continued to be improved or improved further at 2-month and 12-month reassessments. Mean pCO2 increased from hypocapnic to normocapnic levels over the course of treatment and remained normocapnic at follow-up.
«Repeated elevation of pCO2 during homework sessions may have desensitized a hypersensitive suffocation alarm system (Klein, 1993), reducing panic vulnerability. Such desensitization could increase tolerance for incidental increases of arterial pCO2during daily life and result in fewer compensatory hyperventilatory episodes.»
Alternatively, in so far as hyperventilation itself can cause panic attacks (Ley, 1985), practiced skill at raising pCO2 could directly reduce risk. The fact that an inability to normalize breathing quickly after paced hyperventilation was associated with less clinical recovery suggests that respiratory and clinical outcomes were linked.
Thus, non-respiratory mechanisms may also have played a role in patient improvement. For instance, the treatment rationale provided to patients included cognitive components that may have counteracted catastrophic thinking and given patients a greater sense of control. The paced breathing exercises, which often triggered uncomfortable sensations similar to those experienced during panic attacks (Meuret et al., 2003), may have produced interoceptive exposure and desensitization to bodily cues that was not respiration-specific (Craske et al., 1997). The slight decreases of pCO2 during home-exercises could be indicative of such an unpleasant exposure effect.