Nevner hvordan betennelsesmarkører og insulinsensitivitet henger sammen. Svært mange viktige poenger og en god oversiktig i denne studien. Info om betennelser, insulin, tyroksin, fett, oksidativt stress, D-vitamin, m.m.
http://www.jnutbio.com/article/S0955-2863(13)00054-5/abstract
http://www.jnutbio.com/article/S0955-2863(13)00054-5/fulltext
Fra evolusjonens side er insulinresistens en overlevelsesmekanisme. Hjernen og nervesystemet bruker 20% av energien glukose metabolismen gir oss og for overlevelse er hjernen førsteprioritet. Når det er betennelser i kroppen vil immunforsvaret naturlig bruke opp mye av glukosen i blodet slik at det går på bekostning av hjernens energitilgang. Da må hjernen sette igang en «insulinresistens» for å sørge for at insuline ikke fjerner glukosen i blodet. Insulinresistens er en livsviktig mekanisme som skal fungere i kort tid av gangen (noen dager). Om det blir en kronisk systemisk betennelse blir det store problemer i kroppen som etter mange år gir utslag i livsstilsykdommer.
«Our sensitivity to develop insulin resistance traces back to our rapid brain growth in the past 2.5 million years. An inflammatory reaction jeopardizes the high glucose needs of our brain, causing various adaptations, including insulin resistance, functional reallocation of energy-rich nutrients and changing serum lipoprotein composition. «
Betenneler er en naturlig helbredelsesreaksjon ved skade eller infeksjoner. Men i vår vestlige kultur har vi introdusert en rekke «falske» betennelsestriggere. Altså elementer som gir kroppen betennelser uten at det foreligger en skade. Når betennelser blir kroniske er det ett eller annen «kronisk» vi gjør i vår livsstil som opprettholder betennelsene. Maten vi spiser er den viktigste bidragsyteren spesielt siden vårt moderne kosthold inneholder så mange betennelsestriggere. Bl.a. sukker og mel og overvekt av raffinerte næringsfattige karbohydrater. Skal man bli frisk fra kroniske plager og betennelser i muskel- og skjelettapparatet må man fjerne de falske betennelsestriggerene fra hverdagen.
«With the advent of the agricultural and industrial revolutions, we have introduced numerous false inflammatory triggers in our lifestyle, driving us to a state of chronic systemic low grade inflammation that eventually leads to typically Western diseases via an evolutionary conserved interaction between our immune system and metabolism. The underlying triggers are an abnormal dietary composition and microbial flora, insufficient physical activity and sleep, chronic stress and environmental pollution. «
Betenneler blir viktigere og viktigere i helsesammenheng. Hjerte/kar problemer, flere kreftformer, degenererende sykdommer, m.m. har alle samme utgangspunkt: systemisk betennelse og medfølgende insulinresistens.
«In recent years, it has become clear that chronic systemic low grade inflammation is at the basis of many, if not all, typically Western diseases centered on the metabolic syndrome. The latter is the combination of an excessive body weight, impaired glucose homeostasis, hypertension and atherogenic dyslipidemia (the “deadly quartet”), that constitutes a risk for diabetes mellitus type 2, cardiovascular disease (CVD), certain cancers (breast, colorectal, pancreas), neurodegenerative diseases (e.g., Alzheimer’s disease), pregnancy complications (gestational diabetes, preeclampsia), fertility problems (polycystic ovarian syndrome) and other diseases [1]. Systemic inflammation causes insulin resistance and a compensatory hyperinsulinemia that strives to keep glucose homeostasis in balance. Our glucose homeostasis ranks high in the hierarchy of energy equilibrium, but becomes ultimately compromised under continuous inflammatory conditions via glucotoxicity, lipotoxicity, or both, leading to the development of beta-cell dysfunction and eventually Type 2 diabetes mellitus [2].»
Det er en evolusjonært tilpasset sammenheng mellom kroppsvekt og metabolisme hos dyr. Jo større dyret er jo mer energi krever metabolismen. Hjernen og nervesystemet er et av de mest energikrevende organene så når mennesket etterhvert utviklet en enorm hjerne relativt til kroppsvekt måtte dette gå på bekostning av andre energikrevende organer. Hos oss har tarmene blitt mindre.
«Our brain consumes 20–25%2 of our basal metabolism [11], [12], [13], [14], [15], [16], [17] and [20] and is thereby together with the liver (19%2), our gastrointestinal tract (15%2), and skeletal musculature (15%2) among the quantitatively most important organs in energy consumption [19].»
«There is a linear relationship between body weight and basal metabolism among terrestrial mammals (Fig. 2). This apparently dogmatic relationship predicts that, due to the growth of our brain, other organs with high energy consumption had to be reduced in size, what in evolution is known as a “trade-off”.3 As a consequence of this “expensive tissue hypothesis” of Aiello and Wheeler [19], our intestines, amongst others, had to become reduced in size. «
Når vi får mange på glukose i blod blir det konkurranse mellom organene om å få nok. Dette skjer bl.a. under faste, i graviditet og under infeksjoner og betennelser. Hjernen vil alltid være førsteprioritet. Derfor har evolusjonen utviklet insulinresistens for å sørge for at glukose alltid er tilgjengelig for hjernen, uansett hvor mye andre organer eller immunforsvar prøver å ta det.
«A glucose deficit leads to competition between organs for the available glucose. As previously mentioned, this occurs during fasting, but also during pregnancy and infection/inflammation. «
«During competition between organs for glucose, we fulfill the high glucose needs of the brain by a reallocation of the energy-rich nutrients, and to that end, we need to become insulin resistant.»
Insulinresistens påvirker også blodgjennomstrømning ved at det hemmer vasodilatsjon (utvidelse av blodkar). Når blodårene blir trangere må hjertet pumpe hardere og vi får høyt blodtrykk. I tillegg vil manglende fleksibilitet i blodårene gjøre at nyrene blir veldig sensitive for salt. Nyrene kan hjelpe i kontrollen av blodtrykk ved å senke eller øke saltmengden, men dette er en sekundær funksjon. Det er blodkarenes utvidelse og sammentrekning som er er førsteprioritet i blodtrykkskontroll. Når denne funksjonen blir dårlig må nyrene jobbe på høygir og blir etter hvert overarbeidet og skadet.
» For example, the concomitant hypertension has been explained by a disbalance between the effects of insulin on renal sodium reabsorption and NO-mediated vasodilatation, in which the latter effect, but not the first, becomes compromised by insulin resistance, causing salt sensitivity and hypertension [54].»
Her er en gjennomgang av alle aspektene og mekanismene i kroppen som påvirkes av lav-grads betennelser og medfølgende insulinresistens.
«However, it becomes increasingly clear that we could better refer to it as the “chronic systemic low-grade inflammation induced energy reallocation syndrome”. The reason for this broader name derives from the recognition that insulin resistance is only part of the many simultaneously occurring adaptations. To their currently known extent, these adaptations and consequences are composed of:
(i) reduced insulin sensitivity (glucose and lipid redistribution, hypertension),
(ii) increased sympathetic nervous system activity (stimulation of lipolysis, gluconeogenesis and glycogenolysis),
(iii) increased activity of the HPA-axis [hypothalamus-pituitary-adrenal gland (stress) axis, mild cortisol increase, gluconeogenesis, with cortisol resistance in the immune system],
(iv) decreased activity of the HPG-axis (hypothalamus-pituitary-gonadal gland axis; decreased androgens for gluconeogenesis from muscle proteins, sarcopenia, androgen/estrogen disbalance, inhibition of sexual activity and reproduction),
(v) IGF-1 resistance (insulin-like growth factor-1; no investment in growth) and vi) the occurrence of “sickness behavior” (energy-saving, sleep, anorexia, minimal activity of muscles, brain, and gut) [3].»
Mennesket er det dyret med størst hjerne. Og siden hjernen er stappet full av nervetråder som krever svært mye energi for å fungere blir menneskekroppen svært sensitiv for glukose. Hvis vi får lite glukosetilgang har kroppen en etablert en robuste tilpasningsmekanismer for å overleve, men dette gjelder kun i kortere perioder.
«Summarizing thus far, we humans are extremely sensitive to glucose deficits, because our large brain functions mainly on glucose. During starvation, pregnancy and infection/inflammation, we become insulin resistant, along with many other adaptations. «
De forskjellige tilpasningsmekanismene vi har for å overleve ved betennelsestilstander (inkl insulinresisten) viser oss hvor tett immunsystemet og metabolismen er koblet i kroppen vår. Det er ikke to forskjellige systemer, men vevet inn i hverandre.
«The metabolic adaptations caused by inflammation illustrate the intimate relationship between our immune system and metabolism. This relation is designed for the short term. In a chronic state it eventually causes the metabolic syndrome and its sequelae. We are ourselves the cause of the chronicity. Our current Western lifestyle contains many false inflammatory triggers and is also characterized by a lack of inflammation suppressing factors. These will be described in more detail below.»
Denne studien nevnte tidligere at betennelser skaper insulinresisten som følge av en naturlig overlevelsesmekanisme ved skader og akutte sykdommer. Men i vår moderne hverdag har vi en lang rekke «falske betennelsesfaktorer» som gir oss betennelser uten at det foreligger skade. Her er en oversikt over de viktigste «falske betennelsefaktorer» vi må se opp for om vi ønsker å bli kvitt smerter og livsstilssykdommer:
«Among the pro-inflammatory factors in our current diet, we find:
– the consumption of saturated fatty acids [82] and industrially produced trans fatty acids [83] and [84], a high ω6/ω3 fatty acid ratio [85], [86] and [87],
– a low intake of long-chain polyunsaturated fatty acids (LCP) of the ω3 series (LCPω3) from fish [88] and [89],
– a low status of vitamin D [90], [91] and [92], vitamin K [93] and magnesium[94], [95] and [96],
– the “endotoxemia” of a high-fat low-fiber diet [97] and [98],
– the consumption of carbohydrates with a high glycemic index and a diet with a high glycemic load [99] and [100],
– a disbalance between the many micronutrients that make up our antioxidant/pro-oxidant network [101], [102] and [103], and
– a low intake of fruit and vegetables [103] and [104].
The “dietary inflammation index” of the University of North Carolina is composed of 42 anti- and proinflammatory food products and nutrients. In this index, a magnesium deficit scores high in the list of pro-inflammatory stimuli [105]. Magnesium has many functions, some of them, not surprisingly, related to our energy metabolism and immune system, e.g., it is the cation most intimately connected to ATP [95].
Indirect diet-related factors are
– an abnormal composition of the bacterial flora in the mouth [106], gut [106] and [107], and gingivae [108], [109] and [110].
– Chronic stress[111] and [112],
– (passive) smoking and
– environmental pollution [77],
– insufficient physical activity [113],[114], [115], [116], [117] and [118] and
– insufficient sleep [119], [120], [121], [122] and [123] are also involved.»
«Diets high in refined starches, sugar, saturated and trans fats, and low in LCPω3, natural antioxidants, and fiber from fruits and vegetables, have been shown to promote inflammation [82], [83], [84], [129],[130] and [131] (Table 1).»
«Molecular oxygen is essential to aerobic life and, at the same time, an oxidizing agent, meaning that it can gain electrons from various sources that thereby become “oxidized,” while oxygen itself becomes “reduced”[252] and [253]. In general terms, an antioxidant is “anything that can prevent or inhibit oxidation” and these are therefore needed in all biological systems exposed to oxygen [252].»
«The emergence of oxygenic photosynthesis and subsequent changes in atmospheric environment [254] forced organisms to develop protective mechanisms against oxygen’s toxic effects [255]. »
» Damage by oxidation of lipids[262], [265] and [266], nucleic acids and proteins changes the structure and function of key cellular constituents resulting in the activation of the NFκB pathway, promoting inflammation, mutation, cell damage and even death [252], [260] and [267] and is thereby believed to underlie the deleterious changes in aging and age-related diseases [102] and [244].»
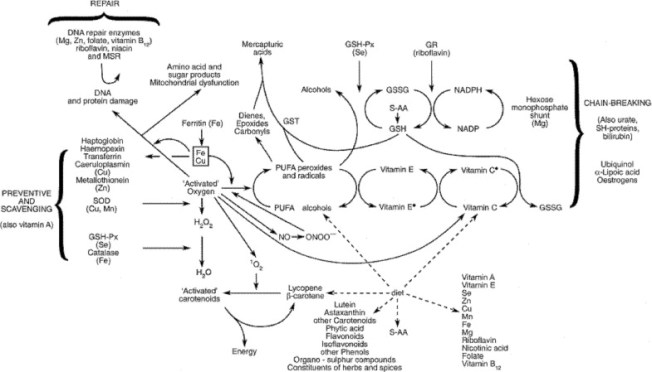
»
Fig. 8. Antioxidant defense mechanisms. An overview of the antioxidant system present in the human body. Various types of antioxidant systems have developed through time, reflecting different selection pressures. Different forms have developed for the same purpose, for example, SODs, peroxidases and GPx are important members of the antioxidant enzyme capacity group. Tocopherols and ascorbic acid, as representatives of the antioxidant network, are manufactured only in plants, but are needed by animals. Ascorbic acid is an essential antioxidant, but cannot be synthesized by Homo sapiens. In humans, therefore, antioxidant defense against toxic oxygen intermediates comprises an intricate network which is heavily influenced by nutrition. GR, glutathione reductase; GSG, reduced glutathione; GSH-Px, glutathione peroxidase; GSSG, oxidized glutathione; GST, glutathione-S-transferase; MSR, methionine sulphoxide reductase; PUFA, polyunsaturated fatty acids; S-AA, sulphur amino-acids; SH-proteins, sulphydryl proteins; SOD, superoxide dismutase; Fe Cu, transition metal-catalysed oxidant damage to biomolecules.»
«A certain level of ROS may also be essential to trigger antioxidant responses [276].»
«Chronic inflammation results in the chronic generation of free radicals, which may cause collateral damage and stimulate signaling and transcription factors associated with chronic diseases [294] and [295].»
«Our diet is composed of millions of substances that are part of a biological network. In fact, we eat “biological systems” like a banana, a fish or a piece of meat. There is a connection between the various nutrients in these systems. In other words, there is a balance and an interaction that is part of a living organism. »
«As clearly explained by Rose[328]: «If everyone smoked 20 cigarettes a day, then clinical, case–control and cohort studies alike would lead us to conclude that lung cancer was a genetic disease; and in one sense that would be true, since if everyone is exposed to the necessary agent, then the distribution of cases is wholly determined by individual susceptibility”. In other words: “disease susceptibility genes” is a misnomer from an evolutionary point of view.»
«Hemminki et al.[326] stated that “if the Western population was to live in the same conditions as the populations of developing countries, the risk of cancer would decrease by 90%, provided that viral infections and mycotoxin exposures could be avoided”.»
«It has become clear that most, if not all, typically Western chronic illnesses find their primary cause in an unhealthy lifestyle and that systemic low grade inflammation is a common denominator.»

